EGFP LNP mRNA
VectorBuilder's EGFP LNP-mRNA has been sequence optimized, fully QC'ed, and functionally validated. It can be used as a control for both in vivo and in vitro applications. Of note, the encapsulated EGFP IVT mRNA was generated with or without modified nucleotide.
Ordering Information Price Match
Click here to view the amino acid sequence translated from EGFP mRNA >>
Shipping and storage
Our LNP-mRNA products are stored in Tris buffer (pH 7.4) and can be stored at -80°C for up to 6 months. The products are shipped on dry ice and should be avoided for freeze-thaw cycles.
Experimental Validation
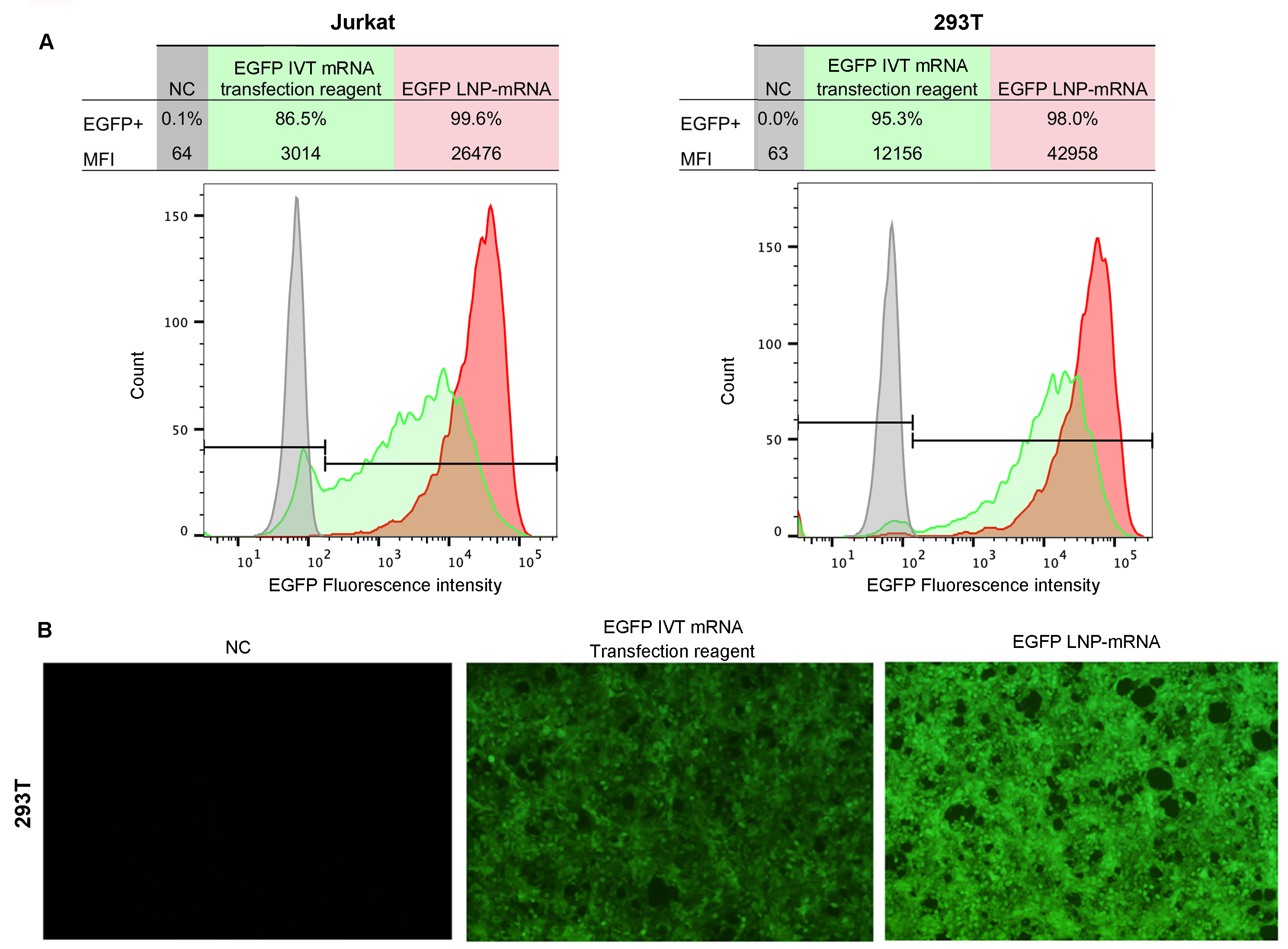
Figure 1. Highly efficient mRNA delivery using LNP
Comparing the cellular expression levels of EGFP mRNA, LNP-encapsulated mRNA demonstrated higher expression compared to EGFP mRNA delivered by a commercial transfection reagent. 1 ug of EGFP mRNA was transfected into cells followed by (A) Flow cytometry analysis of EGFP expression in Jurkat and 293T cells and (B) Fluorescent imaging of 293T cells 24 hours post-transfection. Nucleotides of EGFP mRNA are both unmodified. Median Fluorescence Intensity (MFI).
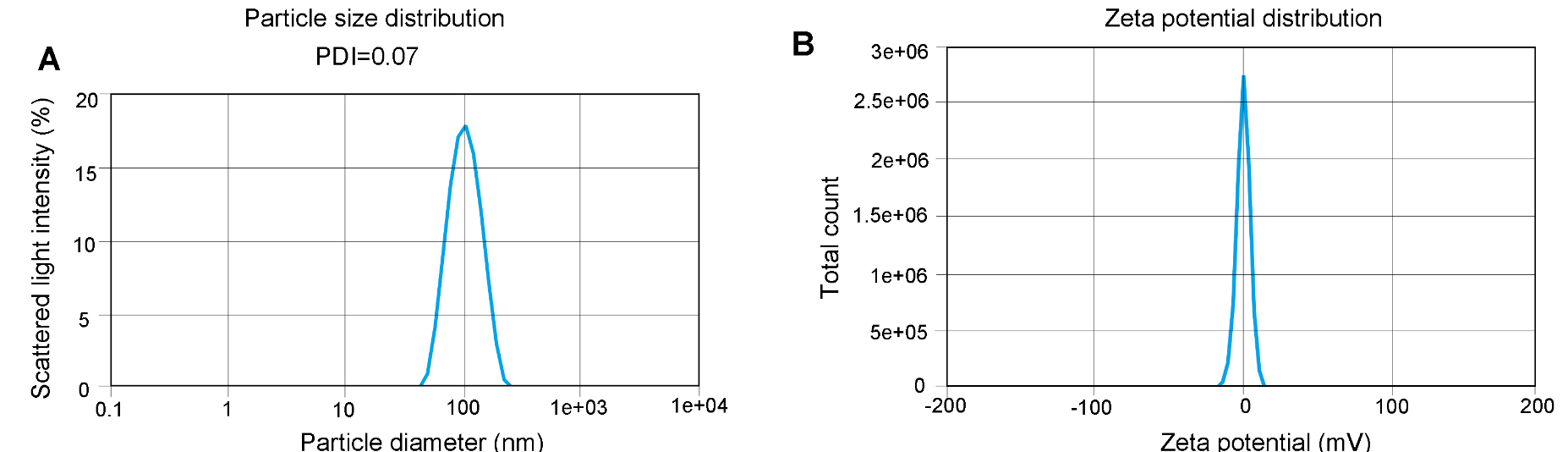
Figure 2. Representative quality control (QC) results of EGFP LNP-mRNA
(A) Particle size was determined by dynamic light scattering (DLS) which measures the intensity differences of fluctuated light due to motion of particles. The polydispersity index (PDI) reflects the heterogeneity of a sample on particle size. (B) Zeta potential reflects the stability of LNP. The Zeta potential of the sample was between -1.872 mV and +1.872 mV.
Documents
Material Safety Data Sheet (MSDS) Certificate of Analysis (COA) User Instructions- IVT mRNA for in vitro applications
- LNP-mRNA for in vitro applications
- LNP-mRNA for in vivo applications
Related Services
IVT mRNA and LNP-mRNAmRNA gene delivery solutions
IVT mRNA and LNP manufacturing
Vector cloning services
Online vector design



