Mammalian Protein Expression
VectorBuilder provides extensive expertise in producing recombinant proteins in mammalian cells. The mammalian recombinant protein expression system enables the target proteins to be expressed in their most natural state, making it the preferred approach for generating therapeutic proteins, vaccines, and antibodies. We offer recombinant protein production in HEK293 cells using a highly optimized transient transfection platform and in CHO cells upon request.
Highlights
- Eukaryotic proteins in their most native state with proper folding
- Contains all necessary post-translational modifications
- Both transient protein expression and stable cell line generation services are available
Service Details
Price and turnaround Price Match
| Service Module | Brief Description | Deliverables | Price (USD) | Turnaround |
|---|---|---|---|---|
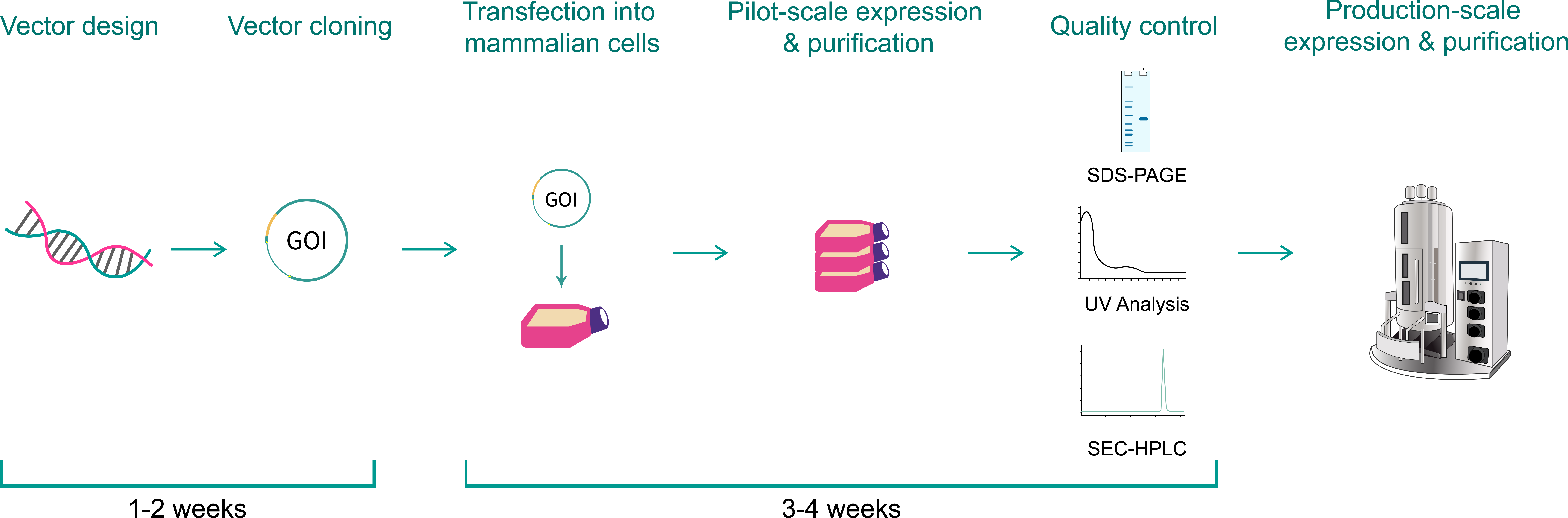
| Vector design and cloning | Our vector design includes fusion partner selection, purification tag selection, and codon optimization. This is followed by gene synthesis and cloning into the mammalian protein expression vector. | E. coli glycerol stock | From $160 | 1-2 weeks |
| Pilot-scale expression and purification | Mammalian cells, such as HEK293 cells, will be transfected. If feasible, 0.1-0.5 mg of purified target proteins * can be produced, and quality control (QC) will be performed. |
|
From $1,800 | 3-4 weeks |
| Production-scale expression and purification | Scaled up production of >100 mg of purified proteins. |
|
Please inquire | |
* Purification methods will be determined based on the construct design and protein characteristics.
Shipping and storage
The default deliverable for vector cloning is E. coli glycerol stock. Upon request, small amounts of plasmid DNA can be provided if available without additional charge. Most of our recombinant proteins are delivered as frozen liquid on dry ice. Upon receipt, they should be stored at -20°C to -80°C under sterile conditions. Recombinant proteins typically remain stable for up to a year if stored properly. Additionally, we recommend aliquoting the recombinant proteins upon receiving and avoiding freeze-thaw cycles.
Quality control (QC)
All vectors cloned by VectorBuilder come with a 100% sequence guarantee. All recombinant proteins produced by VectorBuilder undergo stringent quality control. Default QC for most systems includes 1) validation of the protein expression vector by restriction digestion analysis and Sanger sequencing; and 2) determination of protein concentration and purity by A260/280 measurement and SDS-PAGE. Common additional QC services are shown in the table below, which can be provided upon request.
| Additional QC services | Method |
|---|---|
| Endotoxin test | LAL |
| Protein characterization | Western Blot |
| Intact MS (reduced) | |
| SEC-HPLC | |
| SEC-MALs | |
| Protein N-terminal sequencing | |
| Host cell protein test | |
| Tag removal | Protease digestion |
| Kinetics and affinity analysis | Octet |
| Biacore | |
| Pathogen testing panel for rodents | |
Technical Information
Vector system
Please visit our vector guides on mammalian expression vector systems.
Case study
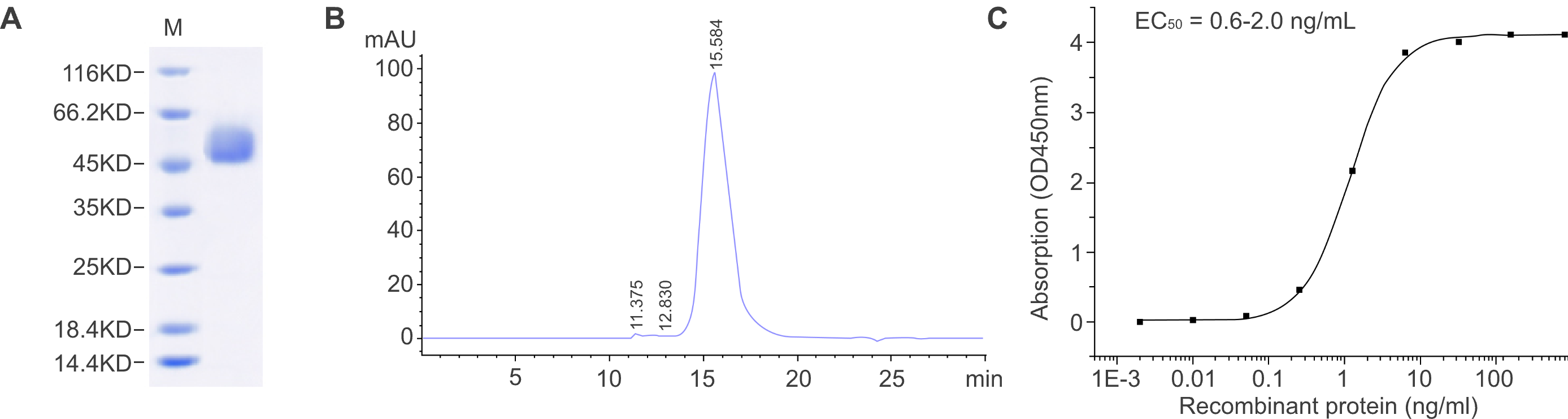
Figure 1. Characterization of a recombinant protein produced in HEK293 cells. (A) SDS-PAGE analysis shows the molecular mass of the recombinant protein, and the purity was determined to be ≥ 95%. (B) The purity was determined to be ≥ 90% by SEC-HPLC. (C) The biological activity of the recombinant protein was measured in an antibody binding assay. The EC50 is between 0.6 and 2.0 ng/ml.
How to Order
Customer-supplied vectors
If customer-supplied materials are needed, please send them to us following the Materials Submission Guidelines. Please strictly follow our guidelines to set up shipment to avoid any delay or damage of the materials. All customer-supplied materials undergo mandatory QC by VectorBuilder, which may incur $100 surcharge for each item. Please note that production may not be initiated until customer-supplied materials pass QC.
Resources
FAQ
All recombinant protein expression systems have their pros and cons which should be taken into consideration while selecting the optimal system for your project. The table below summarizes the pros and cons of each system.
| Recombinant protein expression system | Pros | Cons |
|---|---|---|
| Bacterial |
|
|
| Mammalian cells |
|
|
| Insect |
|
|
| Cell-free system |
|
|
Tags are frequently utilized for recombinant protein production. They streamline the purification process, and certain tags have demonstrated improvement in protein solubility, yield, or purity. If the tag is attached to the protease cleavage site, it can be removed after purification, and the efficiency of cleavage varies depending on the target protein. The careful selection of an appropriate tag is crucial for downstream protein expression and purification. The table below provides an overview of commonly used tags along with their advantages and limitations.
| Tag | Commonly applied system | Advantages | Limitations |
|---|---|---|---|
| GST | Bacteria, insect |
|
|
| His | All |
|
|
| SUMO | Bacteria, insect |
|
|
| Flag | Mammalian cells, insect |
|
|
| MBP | Bacteria, insect |
|
|
| Fc | Mammalian cells, insect |
|
|
For more information regarding tags, please visit protein tags.
For transient transfection, we recommend HEK293 (Human Embryonic Kidney 293) as the first choice. HEK293 cells are easy to transfect, with well-established and efficient procedures that save you time and money. Furthermore, HEK293 cells are easily passaged and maintained in R&D labs and exhibit high levels of protein expression.
In contrast, CHO (Chinese Hamster Ovary) cells pose some transfection challenges. However, they are more suitable for the biopharmaceutical industry, particularly in the production of therapeutic antibodies or proteins. More than 70% of biotherapeutics are manufactured using this cell line.