Gene Knockdown Stable Cell Line
VectorBuilder can engineer shRNA knockdown stable cell lines for applications requiring long-term repression of your gene of interest (GOI). To ensure efficient knockdown of your GOI, the target cells are each separately transduced with lentiviruses each carrying one of the three shRNAs we designed for targeting your GOI. The cell line showing the best knockdown efficiency validated by RT-qPCR will be delivered. Additionally, a series of standard QC assays such as sterility tests and mycoplasma detection are performed before releasing the final cell line products.
Highlights
- Permanent integration: lentiviral transduction allows for stable integration into the genome of host cells and all progeny.
- Broad tropism: VSV-G pseudotyped lentivirus is able to bind to surface receptors on most mammalian cells, facilitating efficient transduction of your target cells.
- Efficient down-regulation: the most effective of the 3 knockdown shRNAs is used in delivered cells.
- Rapid turnaround: knockdown cells can be delivered in as fast as 8 weeks starting from vector design.
Service Details
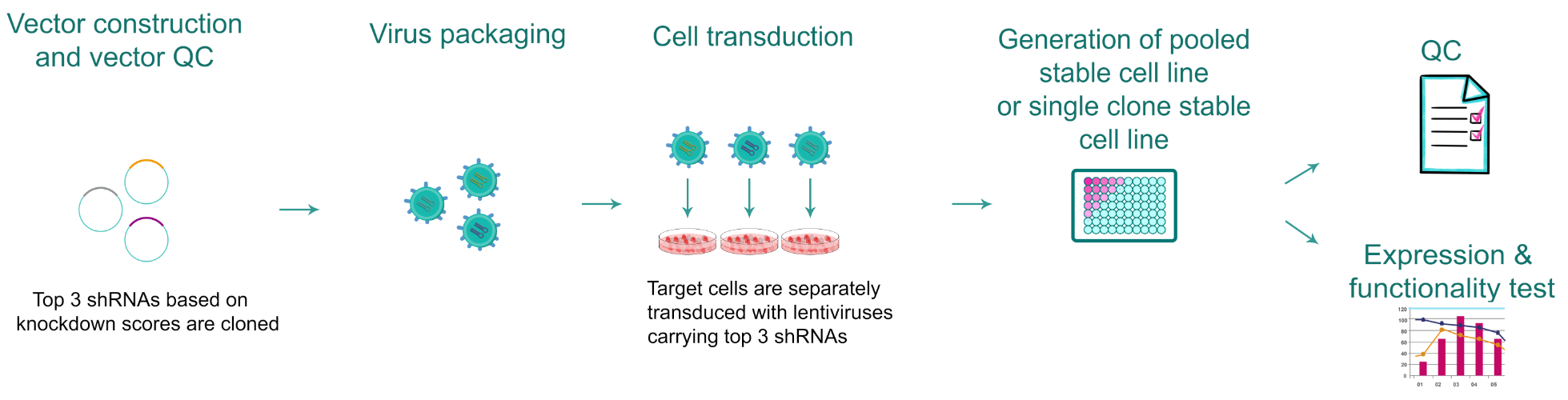
Figure 1. Typical workflow of gene knockdown stable cell line production.
Price and turnaround
| Service Type | Deliverable | Price (USD)* | Turnaround |
|---|---|---|---|
| Gene knockdown | Mixed pool (>106 cells/vial, 2 vials) | From $3,999 | 8-11 weeks |
| Two single clones (>106 cells/vial, 2 vials per clone) | From $4,499 | 13-16 weeks |
* Additional charge will apply for extra single clones or vials.
QC assays
| Assays | Methods |
|---|---|
| Knockdown validation (default) | RT-qPCR |
| Expression test (add-on) | WB, IF, FACS |
| Sterility (default) | PCR for mycoplasma detection, bioburden test for sterility detection |
Downstream services
We can perform various phenotypic assessments and functional validation of your engineered cell lines, including assays for proliferation, apoptosis, migration, viability, cytotoxicity, etc.
Case Studies
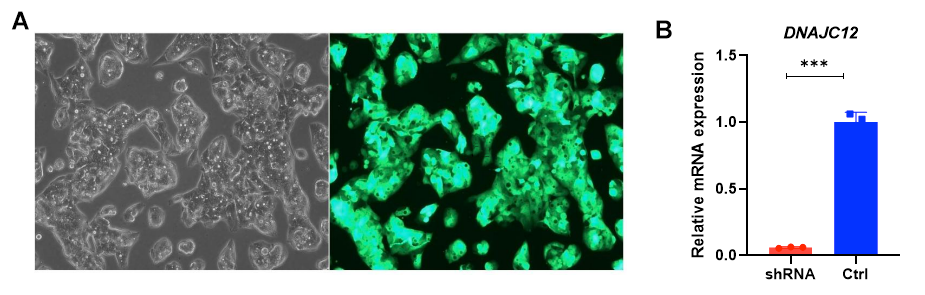
Figure 2. High DNAJC12 knockdown efficiency achieved in Hep-G2 cells. (A) Images of Hep-G2 cells transduced with lentivirus carrying DNAJC12 shRNA with EGFP marker. (B) The knockdown efficiency was 90.9% determined by RT-qPCR. Results were obtained by three repetitions; p value <0.001.
How to Order
Resources
FAQ
VectorBuilder applies rules similar to that used by the RNAi consortium (TRC) to design and score shRNAs. For each given RefSeq transcript, we search for all possible 21mers that are considered as candidate target sites. Candidates are excluded if they contain features thought to reduce knockdown efficiency/specificity or clonability, including a run of ≥4 of the same base, a run of ≥7 G or C, GC content <25% or>60%, and AA at the 5’ end. Knockdown scores are penalized for candidates that contain internal stem-loop, high GC content toward the 3’ end, known miRNA seed sequences, or off-target matches to other genes. For genes with alternative transcripts, target sites that exist in all transcripts are given higher scores.
All scores are ≥0, with mean at ~5, standard deviation at ~5, and 95% of scores ≤15. An shRNA with a knockdown score of about 15 is considered to have the best knockdown performance and clonability, while an shRNA with a knockdown score of 0 has the worst knockdown performance or is hard to be cloned.
Please note that knockdown scores are only a rough guide. Actual knockdown efficiency could depart significantly from what the scores predict. Target sites with low scores may still work well. Also, please note that targeting 3’ UTR can be as effective as targeting coding region.
In order to decide which method is optimal for your specific application, there are a few things you should consider.
Mechanisms
- Knockdown vectors
Knockdown vectors express short hairpin RNAs (shRNAs) that repress the function of target mRNAs within the cell by inducing their cleavage and repressing their translation. Therefore, shRNA knockdown vectors are not associated with any DNA level sequence change of the gene of interest.
- Knockout vectors
CRISPR functions by directing the Cas9 nuclease to cut specific target sites in the genome. These cuts are then inefficiently repaired by the cellular machinery, resulting in permanent mutations, such as small insertions or deletions, at the sites of repair. A subset of these mutations will result in loss of function of the gene of interest due to frameshifts, premature stop codons, etc. If two closely positioned cut sites in the genome (i.e. within several kb) are targeted simultaneously, this can also result in the deletion of the intervening region.
Effectiveness
shRNA-mediated knockdown will never completely repress the expression of the target gene. Even for the most effective shRNAs, some residual expression of the target gene will remain. In contrast, in a fraction of treated cells, CRISPR can generate permanent mutations which may result in complete loss of gene function.
Consistency and uniformity
shRNA vectors generally provide high cell-to-cell uniformity within the pool of treated cells and very consistent results between experiments. In contrast, CRISPR produces results that are highly non-uniform from cell to cell due to the stochastic nature of the mutations introduced. To fully knock out the GOI in a cell, all copies of the gene in the cell must be knocked out. Given that normal cells have two copies of any gene (except for X- or Y-linked genes) while cancer cells can have more than two copies, such full knockout cells may represent a very small fraction of all the treated cells. For this reason, CRISPR knockout experiments require the screening of clones by sequencing to identify the subset in which all copies of the GOI have been knocked out.
Off-target effects
Off-target effects have been reported for both shRNA-mediated knockdown and nuclease-mediated knockout. The off-target phenotype(s) can be estimated by using multiple different shRNAs to target the same gene. If a gene knocked down by multiple different shRNAs results in consistent phenotype(s), then it argues against the phenotype(s) being caused by off-target effects. For CRISPR-mediated knockout, multiple clones containing loss-of-function mutations can be analyzed in order to account for any phenotype(s) that may be due to off-target mutations. Additionally, bioinformatically identified off-target sites could be sequenced in the clones to see if they have been mutated.



